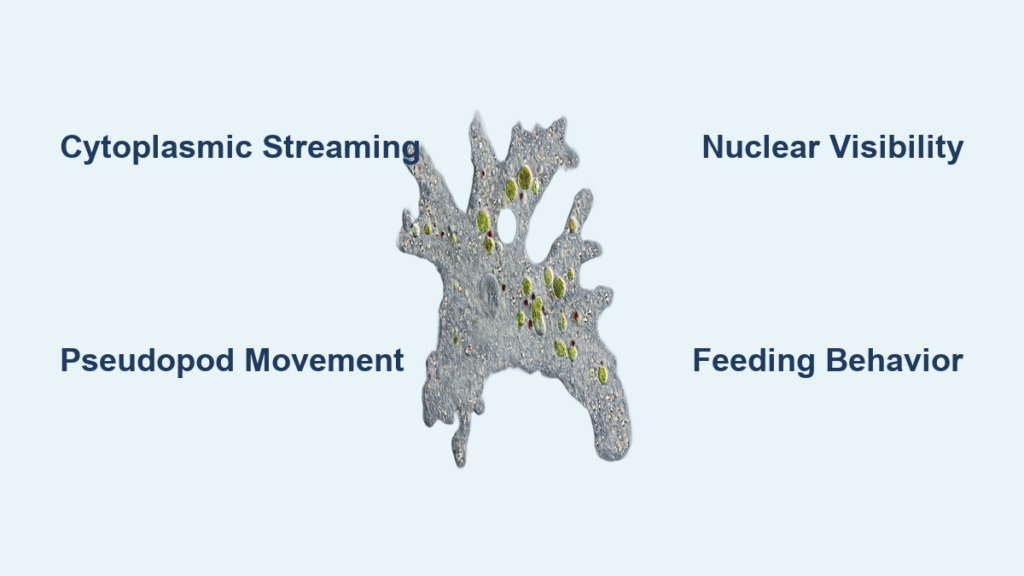
Peering into a drop of pond water under a microscope, you might spot a slow-moving, shapeless form gliding across the field—this is likely an amoeba under microscope. Far from the “simple blob” it’s often dismissed as, the amoeba reveals a remarkably intricate single-celled organism. Using flowing cytoplasm and temporary extensions called pseudopodia, it crawls and feeds in a display of primitive yet highly effective cellular mechanics. These eukaryotic protists offer a captivating window into how life functions at its most fundamental level.
Under magnification, the amoeba’s internal dynamics come alive: streaming cytoplasm, pulsating vacuoles, and rhythmic pseudopod formation. Species like Amoeba proteus and the giant Pelomyxa palustris vary in size and structure but share the defining trait of amoeboid movement. Whether you’re a student, educator, or amateur microscopist, knowing what to look for—and how to find it—can transform a routine slide into a vivid exploration of microscopic survival. This guide walks you through exactly what you’ll see, how to identify key species, and the best techniques for clear, insightful observation.
Key Species to Identify
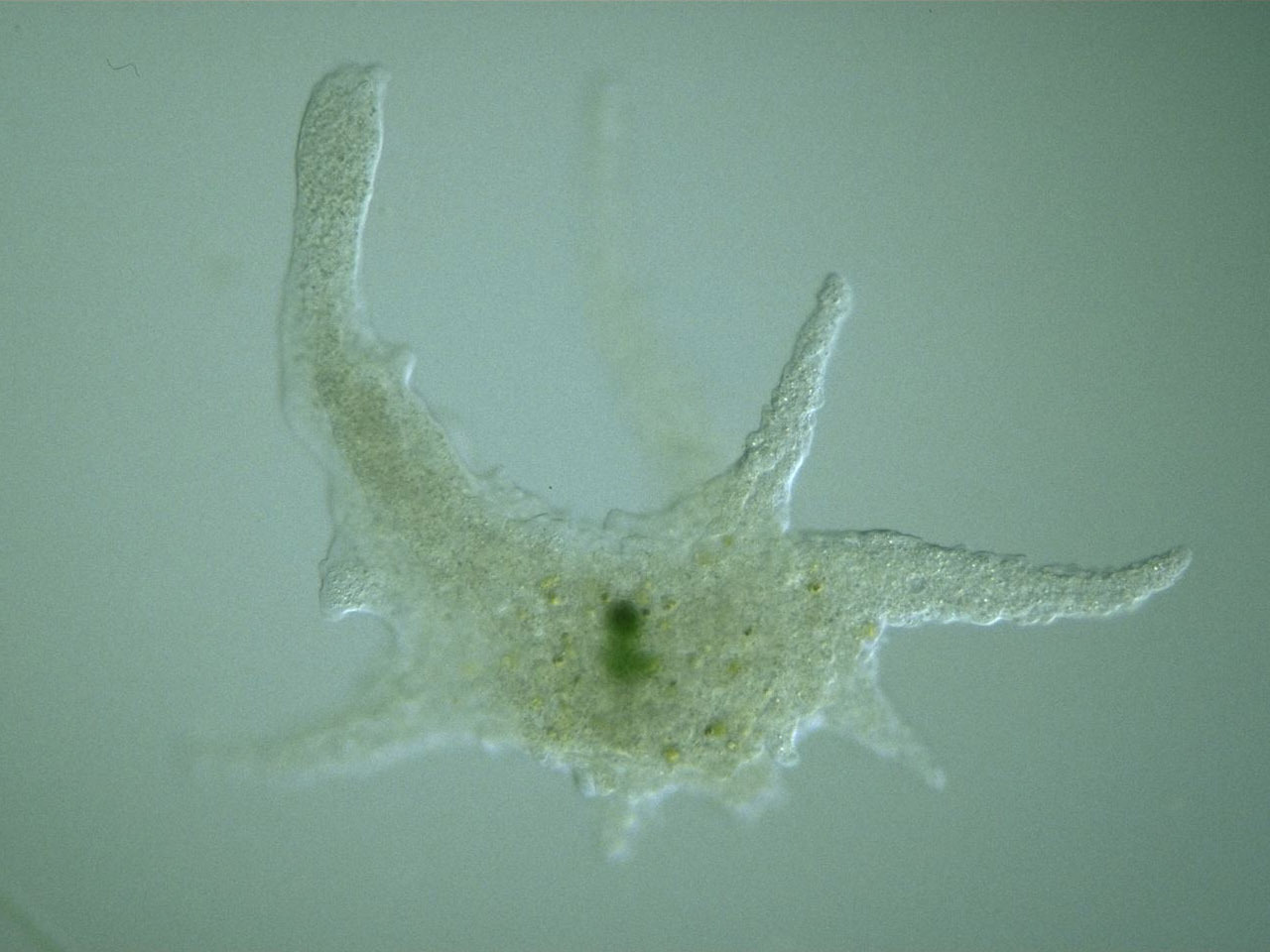
Each amoeba species has distinct microscopic features that aid identification. Recognizing these differences enhances accuracy and deepens observational learning.
Amoeba proteus: The Classic Example
Amoeba proteus is the most commonly observed species in classrooms due to its size and visibility. It reaches up to 0.5 mm—large enough to appear as a tiny speck without magnification. Under the microscope, it appears transparent and gelatinous, with visible cytoplasmic streaming. Its single, dark oval nucleus is clearly visible in stained specimens at 100x magnification. The broad, blunt pseudopodia extend and retract as it moves, engulfing bacteria or algae through phagocytosis. In darkfield illumination, the contrast between the clear ectoplasm and granular endoplasm becomes striking, revealing continuous internal motion. Unlike fast-moving ciliates, A. proteus moves deliberately, making it easy to track in mixed samples.
Chaos carolinensis: Giant With Many Nuclei
Nearly identical in appearance to A. proteus, Chaos carolinensis is often misidentified without close inspection. The key difference? Nuclear count. While A. proteus has one nucleus, Chaos carolinensis contains hundreds. This multinucleate condition supports its slightly larger size and metabolic demands. Definitive identification requires nuclear staining or high-resolution phase-contrast microscopy. Found in the same freshwater sediments as A. proteus, it shares similar movement and feeding behaviors but stands out under proper illumination due to its complex internal structure.
Pelomyxa palustris: Visible Without Magnification
One of the largest known amoebae, Pelomyxa palustris can grow up to 5 mm—visible to the naked eye as a tiny, gelatinous speck. It inhabits oxygen-poor mud in freshwater ponds, thriving where bacteria are abundant. Multinucleated with hundreds of nuclei, it uses its size to dominate microbial communities. Best detected first under a stereo-microscope, Pelomyxa moves slowly and can be isolated from detritus using the inverted jar method. Its transparency and size make it ideal for field collection and classroom demonstration.
Entamoeba histolytica: Human Parasite
Unlike free-living pond amoebae, Entamoeba histolytica is a pathogenic species responsible for amebic dysentery. Transmitted through contaminated water or food, it infects the human intestine and can cause severe diarrhea, abdominal pain, and even liver abscesses. Microscopically, it’s smaller than A. proteus and less active when dormant. Pseudopodia form only during tissue invasion. Diagnosis relies on identifying the organism in stool samples—a critical skill in clinical microbiology, especially in regions with poor sanitation.
Paramoeba spp.: Hosts Microbial Symbionts
Paramoeba species are less commonly seen but scientifically significant. They harbor kinetoplastid endosymbionts within their cytoplasm—microbial partners that may aid metabolism. These amoebae grow slowly in culture, making lab study challenging. Their presence is confirmed through specialized staining or genetic analysis, not just morphology. They serve as models in research on symbiosis and microbial evolution, highlighting the hidden complexity within single-celled life.
How Amoebae Appear Under Different Microscopes

The visibility and detail of an amoeba depend heavily on the microscopy technique used. Each method reveals different aspects of structure and behavior.
Brightfield Microscopy (Stained Slides)
Best for fixed, stained specimens, brightfield microscopy at 100x–400x shows a pink-stained cytoplasm and a dark, oval nucleus. The ectoplasm (outer layer) and endoplasm (inner granular zone) are clearly differentiated. However, this method kills the cell, so motility and feeding cannot be observed. It’s ideal for classroom slides where structure, not behavior, is the focus.
Darkfield Illumination
For live, unstained specimens, darkfield is unmatched. At up to 400x, the amoeba appears bright against a dark background, filled with moving granules—the “confetti effect.” This highlights cytoplasmic streaming, pseudopod formation, and organelle dynamics in real time. It’s perfect for demonstrating natural behavior in teaching labs, allowing students to watch feeding and movement without staining.
Phase-Contrast Microscopy
This technique provides high-detail imaging of live cells without staining. It reveals fine differences in cytoplasmic density, contractile vacuole activity, nuclear membranes, and food vacuoles. Researchers and advanced educators use phase contrast to study physiological processes like osmoregulation and digestion in real time.
Stereo-Microscopy (Low Power)
At 10x–40x magnification, stereo-microscopy is essential for initial detection. It allows field biologists to locate large species like Pelomyxa palustris in pond sediment before transferring them to higher-magnification scopes. Its low power and wide field make it ideal for scanning detritus and isolating active specimens.
Internal Structures Visible Under Microscope
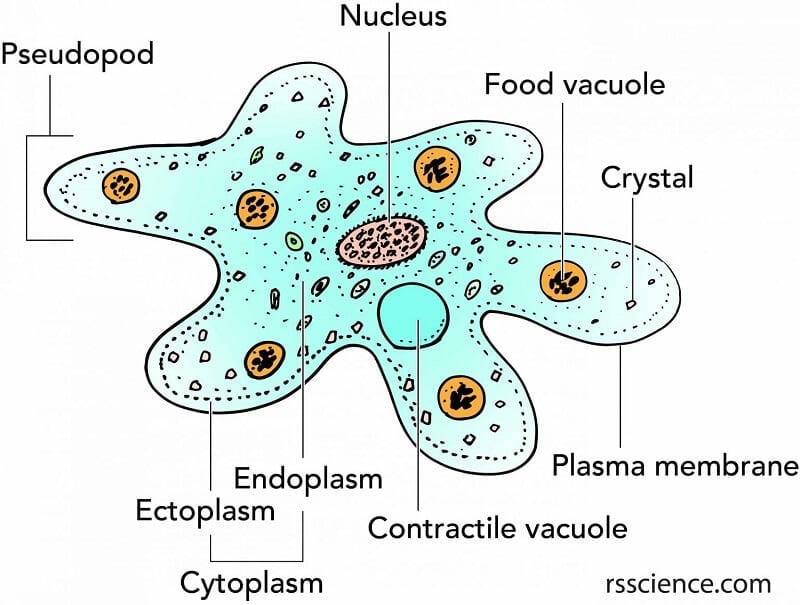
Despite being unicellular, amoebae contain specialized components observable under proper magnification.
Nucleus: Genetic Control Center
The nucleus stores DNA and regulates cell functions. In stained preparations, it’s clearly visible as a dark oval. Species identification often hinges on nuclear count: A. proteus has one; Chaos and Pelomyxa have many. Live-cell staining with DAPI or methylene blue enhances visibility without killing the cell.
Cytoplasm: Two Distinct Layers
The ectoplasm is the clear, gel-like outer layer that provides structure and facilitates movement. The endoplasm is the inner, granular region where organelles and digestion occur. In motion, the boundary between these layers shifts dynamically, visible under darkfield or phase-contrast.
Pseudopodia: Tools for Movement and Feeding
Formed by actin-myosin interactions, pseudopodia enable both locomotion and phagocytosis. Monopodial types extend one dominant pseudopod; polypodial forms use multiple extensions. They flow toward food via chemotaxis and engulf prey by surrounding it.
Vacuoles and Inclusions
Contractile vacuoles expel excess water every 10–30 seconds in freshwater species, maintaining osmotic balance. Food vacuoles form around ingested material, acidify, and fuse with lysosomes for digestion. Crystals may also appear, though their function remains unclear.
How Amoebae Move and Feed
Observing an amoeba under microscope reveals two core biological processes: locomotion and feeding.
Locomotion: Cytoplasmic Streaming in Action
Movement begins with actin polymerization at the leading edge, forming a pseudopod. Liquid endoplasm surges forward, gels into ectoplasm at the rear, and myosin contraction pulls the cell body ahead. This creates a smooth, gliding “oozing” motion, distinct from ciliary or flagellar movement.
Feeding: Phagocytosis Step by Step
- The amoeba detects prey via chemical signals.
- Pseudopodia flow around the microbe, enclosing it.
- A food vacuole forms inside the cell.
- Lysosomes release enzymes to digest the contents.
- Nutrients diffuse into the cytoplasm.
- Undigested waste is expelled through the ectoplasm.
You can observe this by adding stained yeast to a culture and watching under darkfield.
Where to Find Amoebae and How to Collect Them
Target oxygen-depleted pond sediment, moist soil, or organic-rich water. Use the inverted jar method: submerge a jar upside down above sediment, tilt to create suction, and draw in detritus. Let samples settle, screen under stereo-microscopy, and transfer active regions to depression slides.
Common Misidentifications and How to Avoid Them
Amoeba proteus is often mistaken for Chaos carolinensis—only nuclear staining confirms the difference. Transparent amoebae can be confused with debris; look for coordinated cytoplasmic flow and feeding. Smaller species like Entamoeba require 400x magnification and darkfield for detection.
Reproduction: Binary Fission in Real Time
Amoebae reproduce asexually via binary fission. The nucleus divides first, followed by cytoplasmic constriction and cleavage into two daughter cells. This process takes 30–60 minutes and occurs every 1–2 days under optimal conditions.
Why Amoebae Matter: Ecology and Medicine
Ecologically, amoebae regulate bacterial populations and recycle nutrients. Medically, Entamoeba histolytica causes 50 million infections and 100,000 deaths annually. Diagnosis relies on stool microscopy, emphasizing the skill’s global health importance.
Microscopy Tips for Best Results
Use fresh samples, let them rest, and transfer active areas gently. Optimal settings:
– 40x–100x: Locate large species.
– 400x: Study internal details.
– Darkfield/Phase Contrast: Observe live cells.
– Staining: Confirm nuclear count.
Record video to analyze movement. Compare with taxonomic keys. And remember: even experts like Wim van Egmond have struggled to photograph wild A. proteus—patience is key.
Observing an amoeba under microscope transforms a seemingly simple cell into a dynamic system of movement, feeding, and adaptation. With the right techniques, the amoeba becomes more than a blob—it becomes a legend in motion.