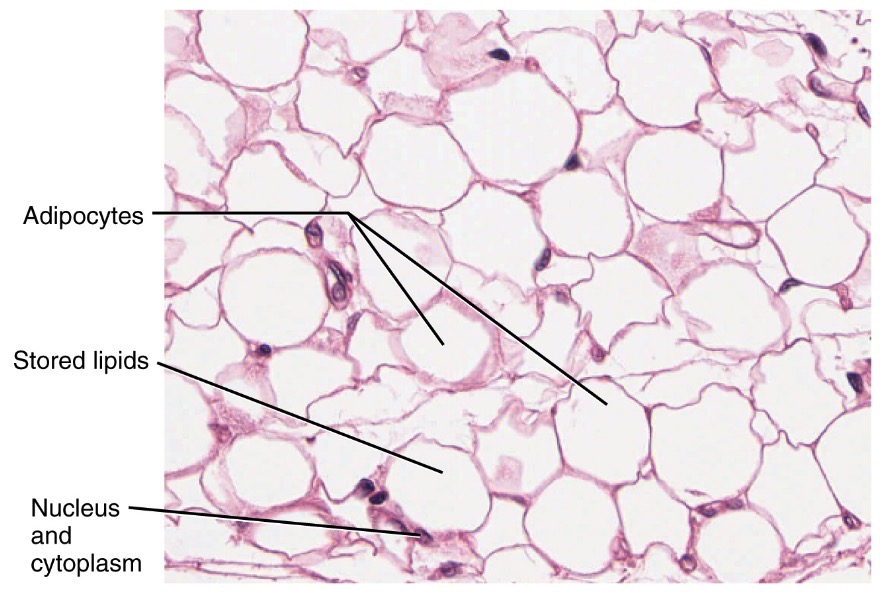
When examining adipose tissue under the microscope, you’re observing one of the most distinctive and easily identifiable tissues in the human body. Adipose tissue under microscope reveals large, empty-looking cells arranged in clusters, with tiny crescent-shaped nuclei pressed against their edges. This unique appearance results from the high lipid content within adipocytes—lipids dissolve during standard histological processing, leaving behind hollow spaces that dominate the field of view.
This visual signature makes adipose tissue a key landmark in histology slides, especially in skin, organ capsules, and connective tissue sections. But beyond its appearance, the microscopic structure tells a story about function: energy storage, insulation, cushioning, and even hormone signaling. Understanding what you’re seeing—whether it’s white or brown fat—reveals critical insights into metabolism and disease.
In this guide, you’ll learn how to identify adipose tissue under the microscope, differentiate between white and brown types, interpret staining results, and understand the clinical significance of what appears as simple “empty space” on a slide.
White Adipose Tissue: Structure and Identification
Large Empty Cells with Peripheral Nuclei
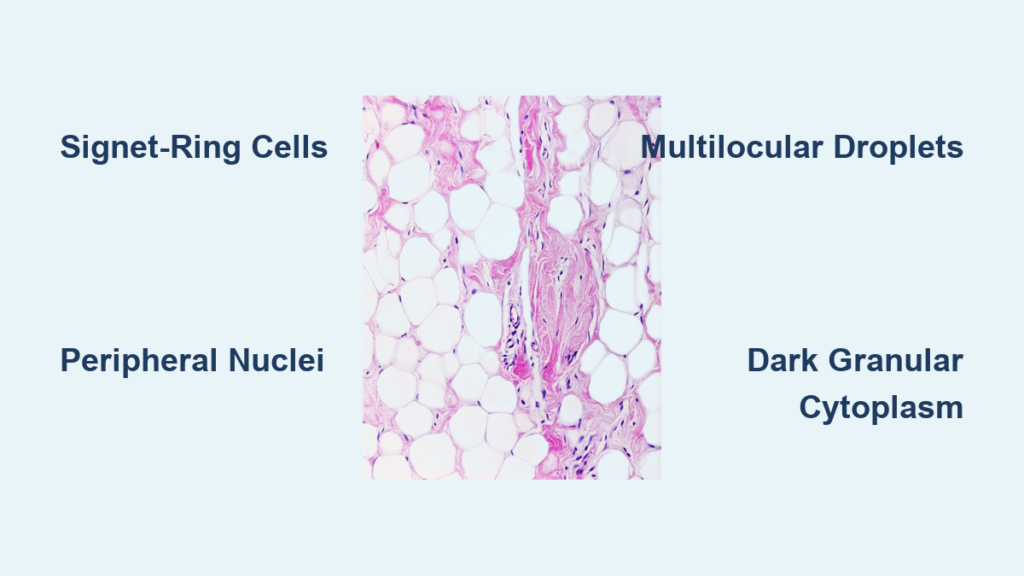
Under light microscopy, white adipose tissue (WAT) appears as vast fields of large, empty, round to polygonal spaces. These are the remnants of adipocytes after lipid extraction during tissue processing. The cytoplasm and nucleus are compressed into a thin rim at the cell periphery, with the nucleus forming a dark, crescent-shaped dot along the edge.
This “signet-ring” morphology is diagnostic for mature unilocular adipocytes. Because lipids dissolve in alcohol and xylene (used in paraffin embedding), no internal structure remains visible—just a clear vacuole surrounded by a faint outline of cytoplasm and a stained nucleus.
The signet-ring appearance is so characteristic that it often serves as a visual anchor in histological sections. When you see these uniformly spaced, circular voids with peripheral nuclei, you can confidently identify the tissue as white adipose, especially in subcutaneous or visceral layers.
Unilocular Lipid Droplets in WAT
Each white adipocyte contains a single large lipid droplet that pushes organelles aside. This unilocular structure maximizes triglyceride storage capacity. In frozen sections stained with Oil Red O, the lipid remains intact and appears bright red or orange, confirming the presence of neutral fats.
On H&E-stained slides, however, these droplets vanish, creating the classic pale, washed-out look. The absence of stain uptake in the central region contrasts sharply with surrounding connective tissue, which stains pink with eosin. This stark contrast helps distinguish adipocytes from other cell types or pathological spaces like cysts.
The size of WAT adipocytes can vary dramatically—from 50 µm in lean individuals to over 100 µm in obesity—reflecting the dynamic nature of fat storage. Larger cells indicate lipid accumulation, while increased cell number suggests hyperplasia, a hallmark of long-term energy imbalance.
Connective Tissue Septa and Stromal Support
Adipocytes aren’t isolated; they’re organized into lobules separated by delicate connective tissue septa. These partitions contain:
– Collagen fibers (type I and III)
– Fibroblasts
– Capillaries (often containing red blood cells)
– Immune cells like lymphocytes and macrophages
These septa provide structural support and vascular supply, essential for nutrient delivery and hormone release. Their presence helps distinguish adipose tissue from artifacts or cystic spaces.
Within the stroma, adipose-derived stem cells (ADSCs) reside, capable of differentiating into adipocytes, bone, or cartilage. This regenerative potential makes adipose tissue a valuable source in regenerative medicine and tissue engineering.
Brown Adipose Tissue: How It Differs Microscopically
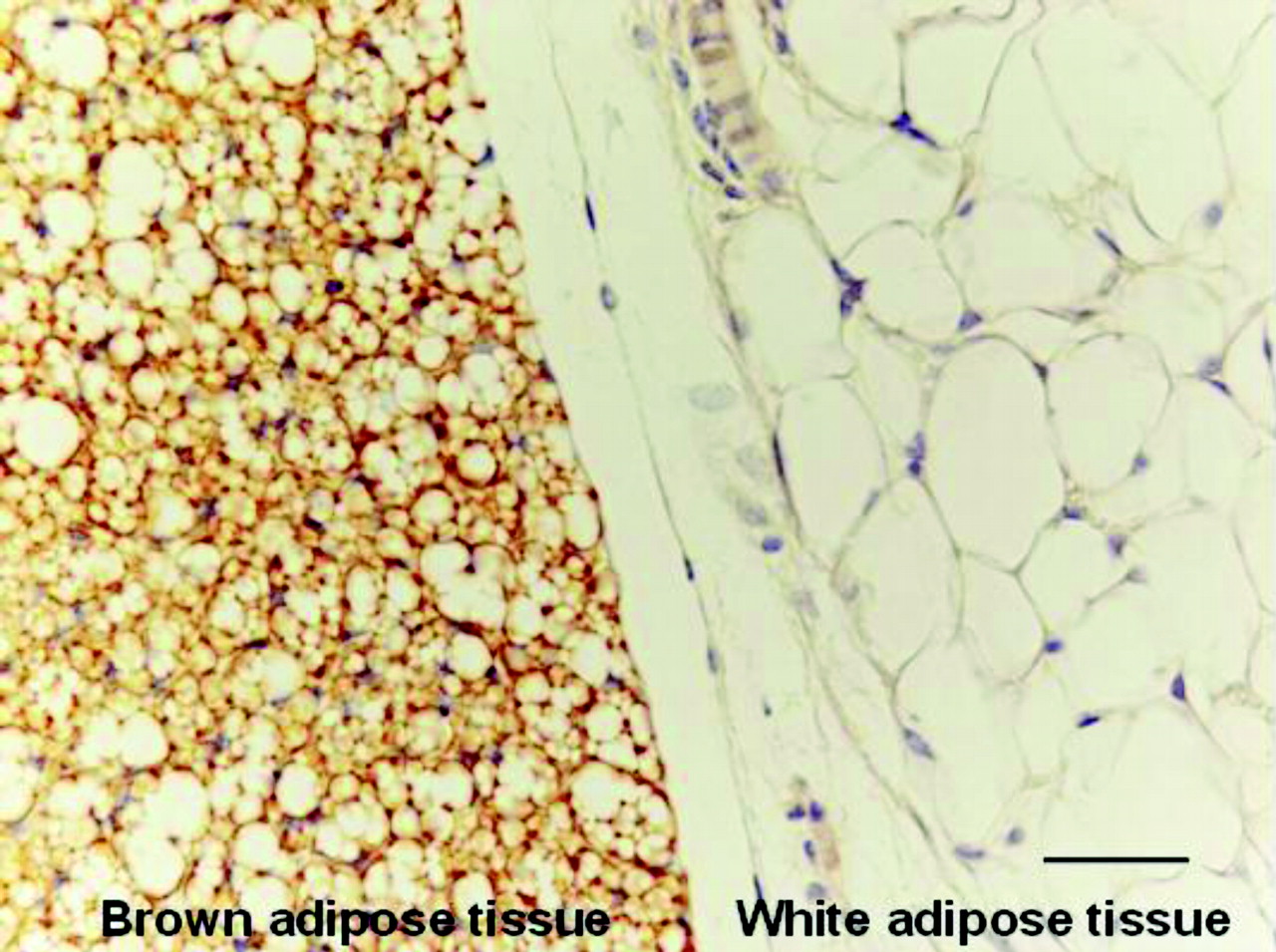
Multilocular Fat Cells with Central Nuclei
Unlike white fat, brown adipose tissue (BAT) has multiple small lipid droplets dispersed throughout the cytoplasm—making it multilocular. This gives the cells a granular, less vacuolated appearance under the microscope.
The nucleus is central, round, and more prominent, not flattened or displaced. Because there’s less lipid per droplet, less is lost during processing, so BAT retains more cellular detail.
This multilocular pattern is a key diagnostic clue. While WAT looks like a field of bubbles, BAT appears more like a cluster of dense, granular cells—each packed with metabolic machinery.
Darker, Granular Cytoplasm Due to Mitochondria
BAT is packed with mitochondria, which contain iron-rich cytochromes that give the tissue a brown color in life—and a darker staining quality in histology. On H&E sections, the cytoplasm appears more basophilic and granular compared to the pale emptiness of WAT.
This high metabolic activity supports thermogenesis via uncoupling protein 1 (UCP1), allowing BAT to burn fat for heat instead of ATP production. This is especially vital in newborns and hibernating animals, where maintaining body temperature is critical.
In adults, active BAT is sparse but detectable in the neck and upper back. Its identification in imaging or biopsy is clinically significant due to its role in energy expenditure.
Limited Distribution in Adults
BAT is abundant in newborns and hibernating animals but sparse in adult humans. When present, it’s found in:
– Neck
– Supraclavicular region
– Upper back
– Paravertebral and periaortic areas
Its identification in biopsies or imaging studies is clinically significant due to its role in energy expenditure and metabolic health. Research into activating BAT is a promising avenue for treating obesity and metabolic syndrome.
Key Differences: White vs Brown Adipose Tissue
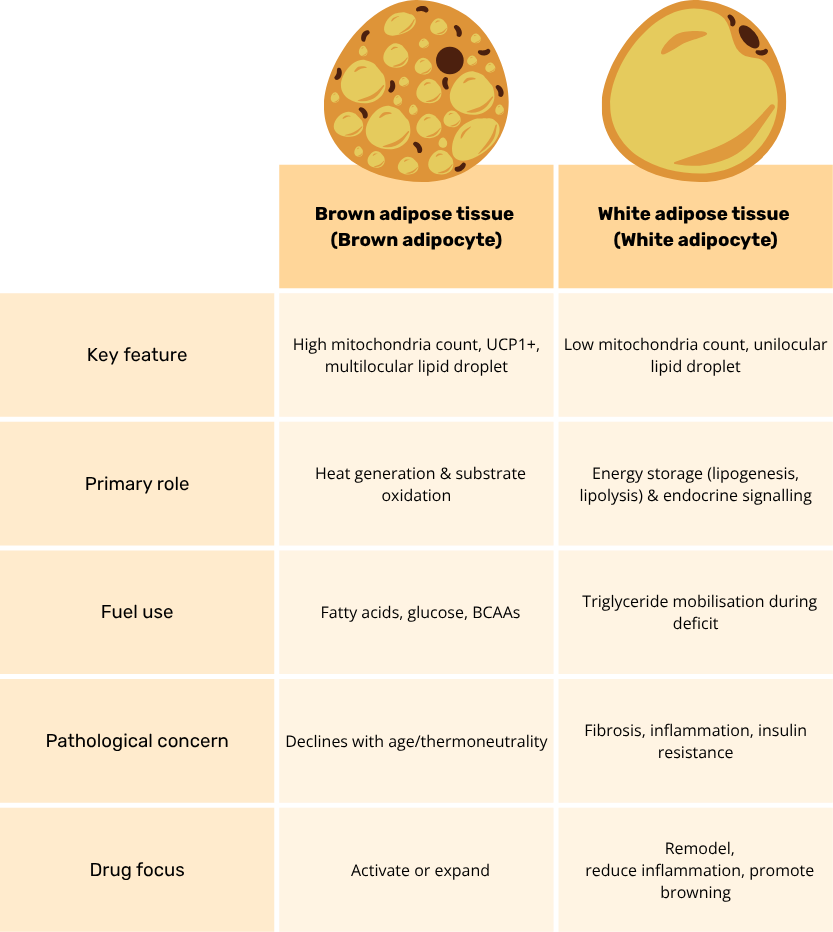
| Feature | White Adipose Tissue (WAT) | Brown Adipose Tissue (BAT) |
|---|---|---|
| Lipid droplets | Unilocular (one large droplet) | Multilocular (many small droplets) |
| Nucleus position | Peripheral, flattened | Central, round |
| Cytoplasm | Thin rim | Abundant, granular |
| Mitochondria | Sparse | Dense, packed |
| Color (in vivo) | White/yellow | Brown |
| H&E appearance | Empty, pale spaces | Darker, cellular, less vacuolated |
| Primary function | Energy storage | Thermogenesis |
| Metabolic rate | Low | High |
These differences make it possible to distinguish the two types even in routine histology, though immunohistochemistry for UCP1 may be used for confirmation in research settings.
Histological Processing: Why Fat Disappears
Lipid Dissolution in Standard Staining
Standard tissue preparation uses formalin fixation, followed by dehydration with alcohol, clearing with xylene, and embedding in paraffin. Since triglycerides are soluble in organic solvents, they dissolve completely during this process.
As a result, lipid droplets vanish, leaving translucent vacuoles in the tissue section. This artifact is universal in paraffin-embedded samples and must be understood to avoid misinterpreting adipocytes as cysts or holes.
How to Preserve Lipids for Microscopy
To visualize actual fat content, special techniques are required:
Use Frozen Sections with Fat-Specific Stains
- Oil Red O: Stains neutral lipids bright red
- Sudan III or IV: Produces orange-red staining
- These stains work best on fresh-frozen tissue, bypassing alcohol dehydration
Alternative Fixatives
- Osmium tetroxide: Preserves lipids and stains them black, useful in electron microscopy
- Toluidine blue (semi-thin sections): Highlights lipid droplets in plastic-embedded tissue
These methods are crucial in research and diagnostic scenarios where lipid quantification is needed, such as in fatty liver disease or lipoma analysis.
Identifying Adipose Tissue: Visual Clues
Look for Signet-Ring Cells
The hallmark of white adipose tissue is the signet-ring cell appearance: an empty space with a dark nuclear crescent at the edge. If you see this pattern across multiple cells, you’re almost certainly viewing adipose tissue.
Check for Lobular Organization
Adipocytes are grouped into lobules separated by thin connective tissue septa. Within these septa:
– Capillaries are common
– Fibroblasts may be visible
– Occasional immune cells (macrophages, lymphocytes) can appear
This organization confirms the tissue is not a fluid-filled cavity or artifact.
Distinguish from Other Tissues
Adipose tissue is rarely confused with other structures, but differential considerations include:
– Cysts or vacuoles: Lack surrounding nuclei and organized lobules
– Foam cells in atherosclerosis: Smaller, intracellular lipid droplets within macrophages
– Clear cell tumors: May have glycogen or lipid, but nuclei are centrally located and disorganized
Context matters—adipose tissue in subcutaneous layers or around organs is expected; its presence in muscle or viscera may indicate ectopic fat deposition, linked to metabolic disease.
Functional Insights from Microscopic Structure
Cell Size Reflects Metabolic State
Adipocyte diameter can reach up to 100 µm when lipid-filled. In obesity:
– Hypertrophy: Existing cells enlarge
– Hyperplasia: New adipocytes form from mesenchymal stem cells
Microscopically, this means larger vacuoles and increased tissue volume. Severe enlargement can lead to hypoxia, fibrosis, and inflammation, visible as immune infiltrates in septa.
Endocrine Activity in the Stroma
Though the adipocyte itself looks inert, the surrounding stroma is metabolically active:
– Leptin, adiponectin, and resistin are secreted by adipocytes
– TNF-α and IL-6 are released by infiltrating macrophages in obese states
These signals influence insulin sensitivity, appetite, and systemic inflammation—linking microscopic findings to whole-body physiology.
Clinical Relevance of Microscopic Findings
Obesity and Visceral Fat Accumulation
Excess white adipose tissue, especially visceral fat, is a hallmark of obesity. Microscopically:
– Adipocytes show hypertrophy
– Septa thicken due to fibrosis
– Crown-like structures (macrophages surrounding dead adipocytes) indicate inflammation
Visceral fat is more metabolically active than subcutaneous fat, releasing more inflammatory mediators and increasing risk for:
– Insulin resistance
– Type 2 diabetes
– Cardiovascular disease
– NAFLD (non-alcoholic fatty liver disease)
Ectopic Fat and Organ Dysfunction
Fat deposition outside normal depots—such as in liver, heart, or skeletal muscle—is detectable microscopically and correlates with organ dysfunction. In fatty liver, hepatocytes contain intracellular lipid vacuoles—similar in appearance to adipocytes but within parenchymal cells.
Summary: Key Identification Criteria
| Feature | Observation Under Microscope |
|---|---|
| Cell shape | Round/polygonal, packed in lobules |
| Vacuoles | Large, empty (WAT); multiple small (BAT) |
| Nucleus | Peripheral, flattened (WAT); central, round (BAT) |
| Cytoplasm | Thin rim (WAT); granular, dense (BAT) |
| Staining (H&E) | Pale, washed-out (WAT); darker, cellular (BAT) |
| Connective tissue | Septa with collagen, vessels, fibroblasts |
| Capillaries | Present in septa |
| Immune cells | Rare in lean; increased in obesity |
Recognizing adipose tissue under microscope goes beyond identifying empty spaces—it’s about understanding the dynamic role of fat in health and disease. From the signet-ring cells of white fat to the granular thermogenic brown adipocytes, each feature reflects a deeper physiological function. Whether you’re a student learning histology or a researcher studying metabolism, mastering the microscopic appearance of adipose tissue unlocks insight into energy balance, endocrine signaling, and the pathology of obesity. With proper technique and attention to detail, what looks like simple “empty space” becomes a window into human metabolic life.