Peering into the hidden world of fungi under the microscope reveals a surprisingly intricate and organized realm far beyond mere mold or mildew. Whether you’re a clinician diagnosing an infection, a plant pathologist identifying a crop disease, or a student exploring microbial life, microscopy unlocks definitive clues about fungal identity and behavior. Unlike bacteria or viruses, fungi display complex, observable structures—hyphae, spores, fruiting bodies—that serve as biological fingerprints. With the right techniques, even a standard compound microscope can distinguish between Aspergillus and Mucor, detect Candida in a smear, or uncover rust spores on a leaf. This guide walks you through the essential steps to confidently analyze fungi under magnification, from slide preparation to final identification—turning visual observation into actionable insight.
Prepare the Sample Correctly
The quality of your microscopic analysis hinges on proper sample preparation. A poorly mounted slide can distort or obscure key features, leading to misidentification.
Choose the Right Mounting Medium
The medium you use affects clarity, preservation, and visibility:
- Water: Quick and accessible, but dries fast and may cause hyphal distortion.
- Lactophenol Cotton Blue (LPCB): The gold standard for fungal mounts—stains chitin blue, slows evaporation, and preserves morphology.
- 85% lactic acid: Excellent for clearing plant tissues, especially when combined with warming.
- 50% glycerol: Reduces drying during prolonged observation, ideal for live or delicate samples.
Seal the edges with nail polish if storing slides long-term—this prevents drying and contamination.
Use Warming to Improve Visibility
Warming softens tough plant cell walls and rehydrates dried spores, dramatically improving clarity:
- Place the slide on a warming plate at 50°C (120°F) for 5–30 minutes.
- A mug warmer is a practical, low-cost alternative.
- Heat helps clear tissues and makes embedded fruiting bodies easier to see.
Avoid overheating—temperatures above 60°C can collapse hyphae or rupture spores.
Transfer Fungal Material Under Dissection Scope
Work under a dissecting microscope (10X–40X) to precisely excise infected tissue:
- Use a scalpel or sterile needle to scrape spore masses or lesioned areas.
- Tap material directly into a drop of mounting medium on the slide.
- Gently lower a coverslip—do not press down hard.
Use oblique lighting during dissection to highlight surface textures and latent fungal growth that might otherwise go unnoticed.
Identify Key Fungal Structures

Accurate identification begins with recognizing diagnostic features of hyphae, spores, and reproductive bodies.
Examine Hyphae First
Hyphae are the building blocks of filamentous fungi. Start by assessing:
- Septate vs. aseptate:
- Septate (e.g., Aspergillus, Fusarium) – cross-walls visible under 40X or 100X.
- Aseptate (coenocytic) (e.g., Mucor, Rhizopus) – broad, ribbon-like filaments without septa.
- Branching pattern:
- Dichotomous branching at 45° – classic for Aspergillus.
- Right-angle branching – common in hyaline molds.
- Irregular, sparse branching – may suggest saprophytic or less aggressive species.
- Hyphal diameter:
- Measure using an ocular micrometer.
- Mucorales: 6–25 μm wide; Aspergillus: 3–6 μm—this difference is a key diagnostic clue.
Analyze Spore Morphology

Spores are the most diagnostic feature. Ask:
What Shape Are the Spores?
- Spherical: Candida, Cryptococcus
- Ellipsoidal: Fusarium, Alternaria
- Fusiform (spindle-shaped): Fusarium oxysporum
- Lemon-shaped: Phytophthora infestans sporangia
- Clavate (club-shaped): Some basidiomycete spores
How Many Septa Do They Have?
- Aseptate (0): Sporangiospores of Rhizopus
- 1-septate: Cladosporium
- 3-septate: Common in Drechslera or Bipolaris
- Multiseptate: Alternaria, Curvularia
Are There Appendages or Sheaths?
- Mucoid sheaths: Pestalotiopsis
- Filamentous appendages: Diplodia conidia
- Hila (scar marks): Important for taxonomic keys
Use oil immersion (100X) to see fine details like septation and surface texture.
Locate Spore Origin Points
Where spores emerge reveals the fungal group:
From Exposed Conidiophores → Hyphomycetes
- Penicillium: Brush-like penicilli with chains of conidia.
- Aspergillus: Conidia radiate from a swollen vesicle.
- Fusarium: Microconidia in false heads; macroconidia sickle-shaped.
From Enclosed Fruiting Bodies → Coelomycetes
- Pycnidia: Flask-shaped structures releasing conidia through an ostiole.
- Example: Phoma on stem cankers.
- Acervuli: Flat, saucer-shaped bodies with dark bristles (setae).
- Example: Colletotrichum causing anthracnose.
From Sporangia → Zygomycetes
- Sporangiophores rise above substrate and end in spherical sporangia.
- When ruptured, release numerous sporangiospores.
- Rhizopus has rhizoids and stolons; Mucor lacks both.
From Asci or Basidia → Sexual Stages
- Asci (sacs) contain ascospores (usually 8 per ascus).
- Look for in Gibberella, Claviceps, or Saccharomyces.
- Basidia produce external basidiospores.
- Found in rusts (Puccinia) and smuts (Ustilago).
Use Stains to Enhance Contrast
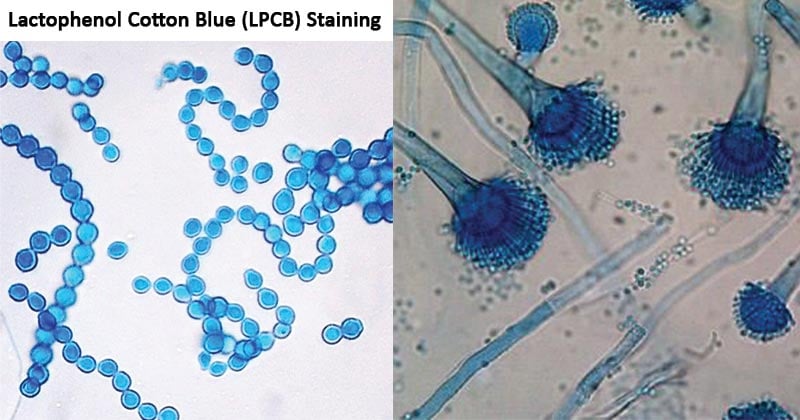
Unstained fungi can be nearly invisible. Specific stains highlight critical features.
LPCB: Go-To Stain for General Use
- Lactophenol Cotton Blue stains chitin in cell walls blue.
- Background clears, improving contrast.
- Ideal for environmental and plant samples.
- Preserves structure for future review.
Calcofluor White for Rapid Screening
- Binds chitin and fluoresces under UV or blue light.
- Detects fungi in clinical specimens quickly.
- Requires fluorescence microscope.
GMS and PAS for Tissue Sections
In histopathology:
- Gomori Methenamine Silver (GMS):
- Fungal walls appear black/silver.
- Gold standard for lung biopsies.
- Periodic Acid-Schiff (PAS):
- Polysaccharides stain magenta-red.
- Excellent for detecting Pneumocystis jirovecii.
India Ink for Capsule Detection
- Used on cerebrospinal fluid (CSF) to diagnose Cryptococcus neoformans.
- Capsule appears as a clear halo around the yeast cell.
- Fast, simple, and highly specific.
Follow a Step-by-Step ID Workflow
A structured approach prevents misidentification and saves time.
Step 1: Determine Host and Context
- Is it a human clinical sample? Plant tissue? Soil isolate?
- Many fungi are host-specific:
- Phakopsora pachyrhizi only infects soybean.
- Candida albicans commonly found in mucosal infections.
- Use host-fungus databases like USDA Fungal Databases or Mycolog.com to narrow options.
Step 2: Observe Under Low Magnification First
Start with 4X or 10X objective:
- Scan for dense hyphal mats.
- Locate fruiting bodies or spore clusters.
- Identify areas rich in reproductive structures.
Switch to 40X or 100X oil immersion for detailed analysis.
Step 3: Measure and Record Critical Dimensions
Use an ocular micrometer to measure:
- Conidia size (length × width in μm)
- Hyphal width
- Number of septa
- Spore wall thickness
Example:
– Fusarium macroconidia: 3–5 septate, 30–60 μm long, fusiform.
– Aspergillus fumigatus conidia: 2–3 μm, spherical to ellipsoidal.
Document all observations—measurements are key for taxonomic keys.
Step 4: Compare to Reference Materials
Match your findings to trusted resources:
For Plant Pathogens:
- Fungi on Plants and Plant Products in the United States (“Big Red Book”) – host-based index.
- Manual of the Rusts in United States and Canada – life cycle diagrams.
- The Genera of Hyphomycetes – illustrated keys.
For Clinical Fungi:
- Atlas of Clinical Fungi (de Hoog et al.)
- CDC fungal identification guides.
Online Tools:
- Mycobank.org: Check current nomenclature and original descriptions.
- Index Fungorum: Verify accepted names and synonyms.
- Google Scholar: Search “host + fungus + microscopy” for recent case studies.
Diagnose Common Pathogens Fast

Recognizing hallmark features speeds up diagnosis in medical and agricultural settings.
Candida albicans: Yeast with Pseudohyphae
- Round to oval yeast cells (3–6 μm), budding.
- Forms pseudohyphae—chains of elongated cells with constrictions.
- Chlamydoconidia visible on cornmeal agar.
- Germ tubes form in serum within 2–3 hours.
Diagnostic clue: Found in oral swabs, vaginal smears, blood cultures.
Aspergillus fumigatus: Septate Hyphae with Acute Branching
- Septate hyphae, 3–6 μm wide.
- Dichotomous branching at 45° angles.
- In culture: Conidiophore with vesicle, phialides, and green conidia.
- Causes invasive aspergillosis in immunocompromised patients.
Stain with GMS or KOH + Calcofluor for confirmation.
Mucor spp.: Broad Aseptate Hyphae
- Wide, ribbon-like hyphae (6–25 μm), no septa.
- Irregular, non-parallel branching.
- Sporangiophores bear apical sporangia filled with spores.
- No rhizoids or stolons (Rhizopus has them).
Linked to mucormycosis—urgent diagnosis required.
Cryptococcus neoformans: Encapsulated Yeast
- Round yeast (4–7 μm) with thick polysaccharide capsule.
- Best seen with India ink in CSF.
- Grows at 37°C; urease-positive.
- Causes meningitis in HIV/AIDS patients.
Avoid Common Microscopy Mistakes
Even experienced users make errors that lead to false IDs.
- Misreading hyphal septation: Aseptate hyphae can appear septate if folded—adjust focus to confirm.
- Confusing bacteria with yeast: Yeast are larger (3–8 μm) than bacteria (0.5–2 μm).
- Overlooking dormant spores: Warm slides to rehydrate spores that may swell or germinate.
- Relying on a single field of view: Examine at least 5–10 fields for consistency.
Final Tips for Success
Microscopy is both science and skill. These practices improve accuracy:
- Start with low magnification—find the “hot spots” first.
- Use oblique lighting during dissection.
- Warm your slides—it makes a visible difference.
- Measure everything—size matters.
- Cross-reference multiple sources.
- Correlate with symptoms—a leaf spot tells you more than spores alone.
While DNA sequencing is powerful, fungi under the microscope remain the fastest, most cost-effective way to get answers—especially in field diagnostics, clinical triage, and education. With practice, anyone can master this essential skill and unlock the hidden world of fungi—one slide at a time.